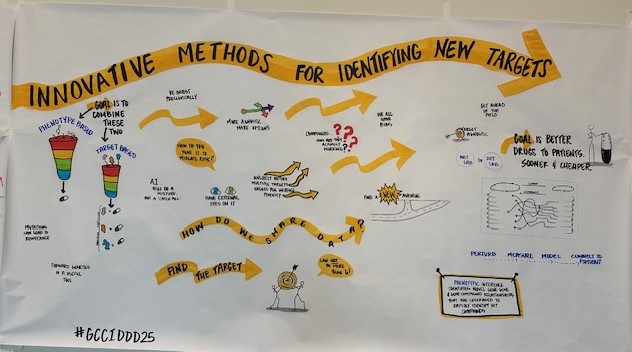
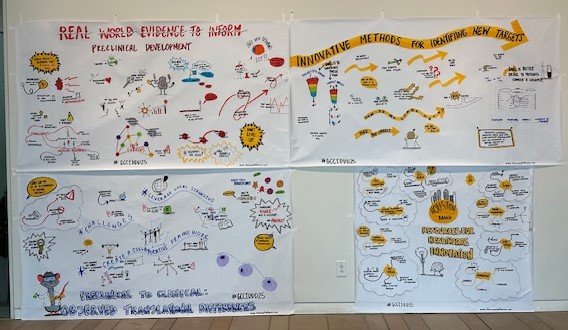
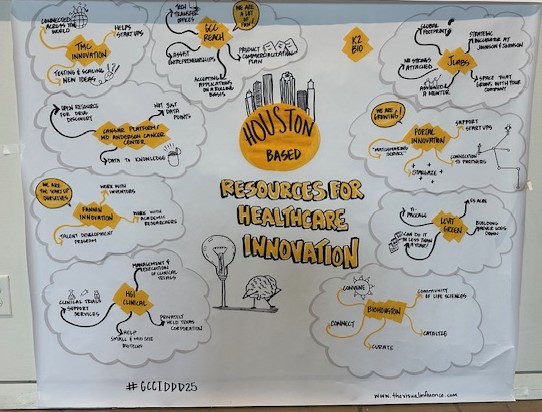
Gulf Coast Consortium for Innovative Drug Discovery and Development
Formerly the John S. Dunn Gulf Coast Consortium for Chemical Genomics (GCC CG) which was formed in 2003, the Consortium for Innovative Drug Discovery and Development (GCC IDDD) is a research consortium focused on providing support for Houston/Galveston scientists in advancing their therapeutic discoveries from inception, through research and development, and to the clinic. IDDD support includes collaborative networking and joint funding opportunities, shared core resources, and educational programs.
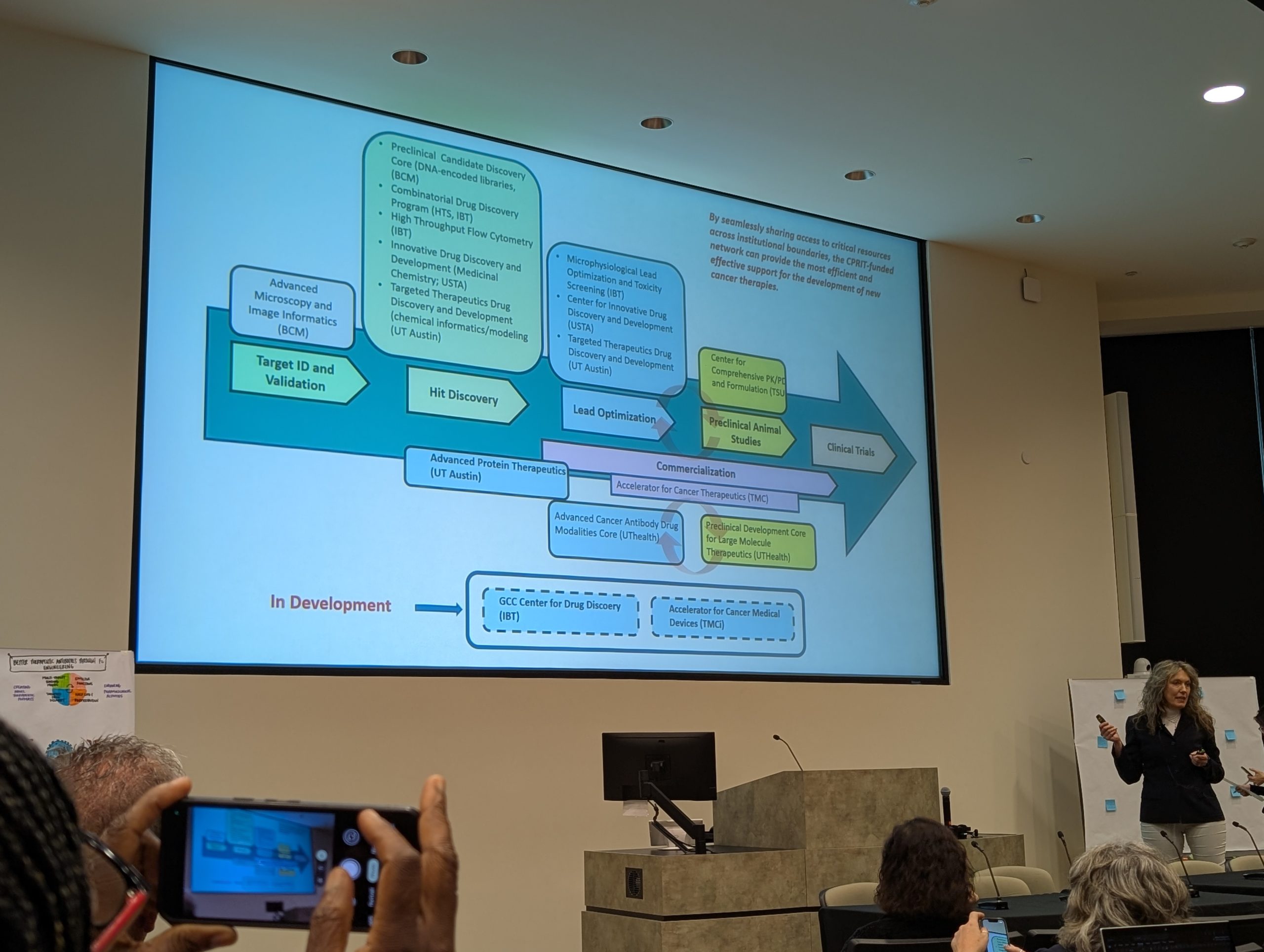
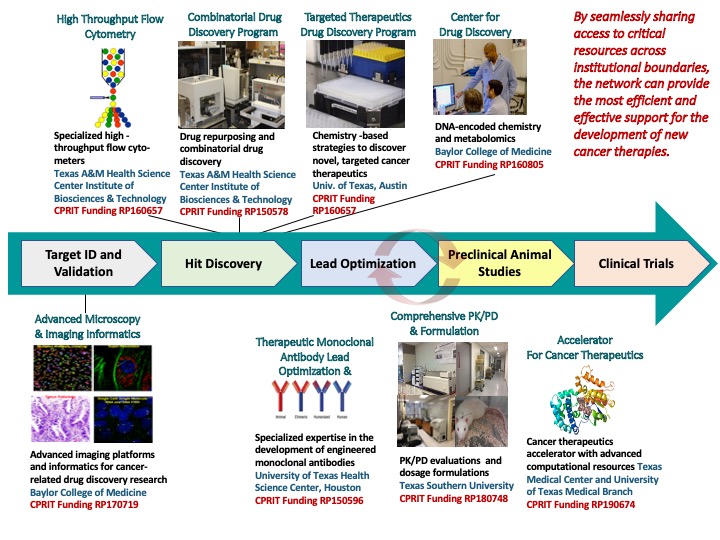
A Statewide Multi-Institutional Model That De-Risks Academic Drug Discovery
The Cancer Therapeutics Training Program (CTTP)
The Cancer Therapeutics Training Program (CTTP) is a multi-institutional post-doctoral training program designed to prepare post-doctoral trainees for future careers in academic and/or commercial cancer therapeutics research and development (R&D). The goal of the program is to recruit and train scientists equipped with the essential skills and knowledge necessary to translate basic cancer research discoveries into commercially viable cancer therapeutics. For more information, visit the CTTP Website.
News
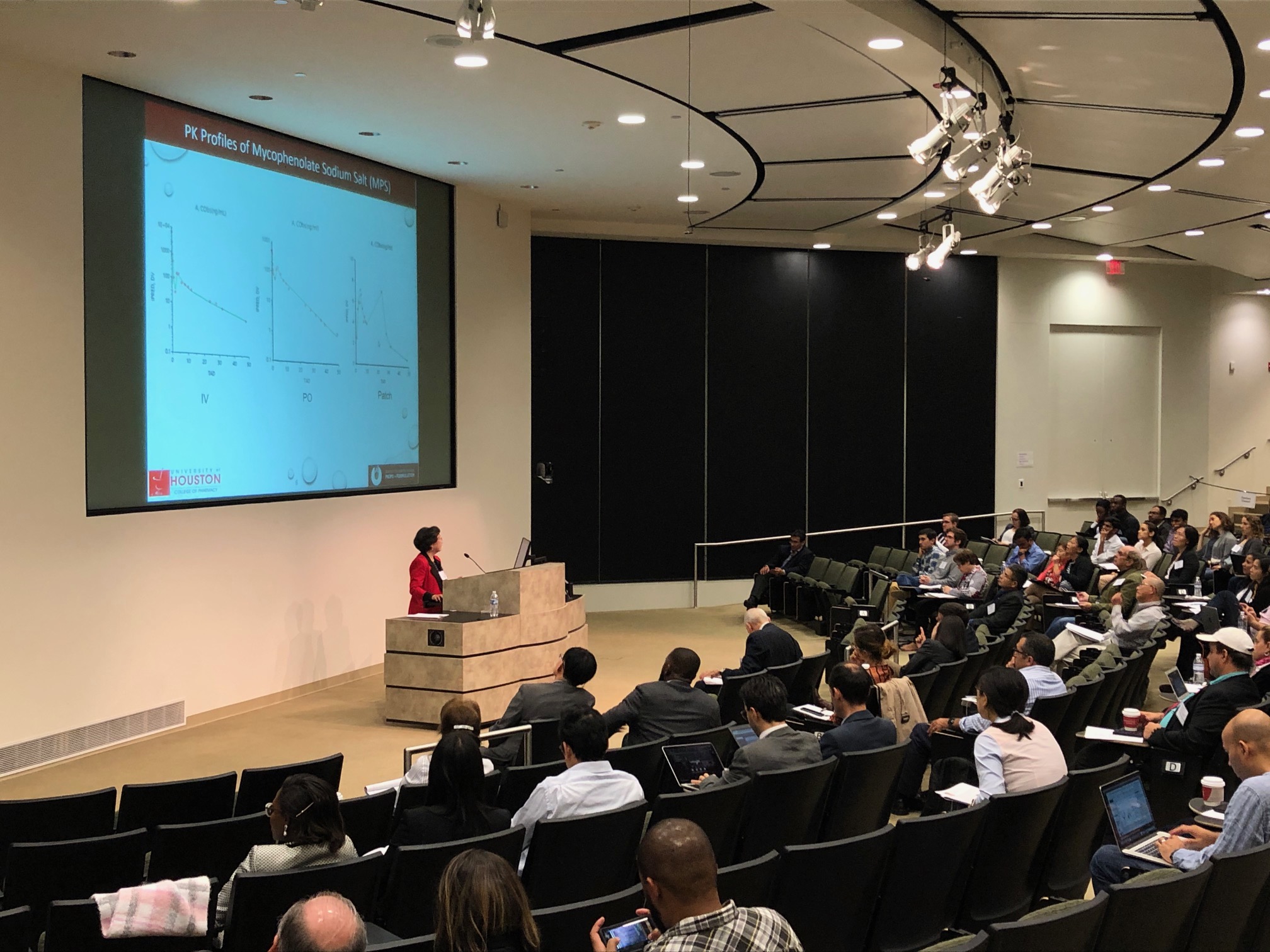
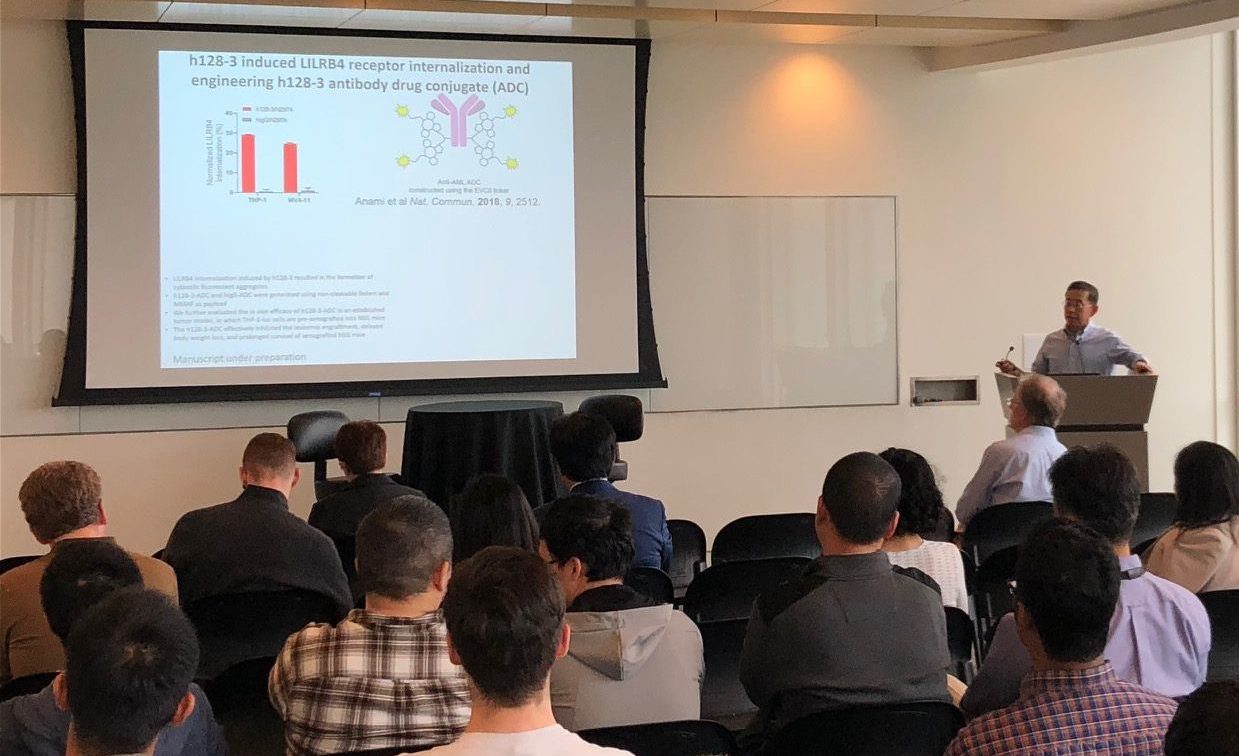
Thank you to everyone who helped make our Kickoff Mini-Symposium a success! For speaker and panel videos, Click Here.
Upcoming Events
There are no upcoming events at this time.
Recent Events
GCC IDDD REACH and BioHouston BioConnect, April 7, 2026
We invite all healthcare technology innovators, entrepreneurs, and biotech stakeholders to join us for the April 7 BioConnect networking series. BioConnect promotes collaboration among academic and industry innovators and key stakeholders within our growing biotech ecosystem. Each month, BioConnect is […]
GCC IDDD REACH and BioHouston BioConnect, March 12, 2026
We invite all healthcare technology innovators, entrepreneurs, and biotech stakeholders to join us for the March 12 BioConnect networking series. BioConnect promotes collaboration among academic and industry innovators and key stakeholders within our growing biotech ecosystem. Each month, BioConnect is […]